Antibody-drug Conjugate Information
General Information of This Antibody-drug Conjugate (ADC)
| ADC ID |
DRG0EPKTP
|
|||||
|---|---|---|---|---|---|---|
| ADC Name |
Trastuzumab-Compound (ld) DAR 1.6
|
|||||
| Synonyms |
Trastuzumab Compound (ld) DAR 1.6
Click to Show/Hide
|
|||||
| Organization |
Orum Therapeutics, Inc.
|
|||||
| Drug Status |
Investigative
|
|||||
| Indication |
In total 2 Indication(s)
|
|||||
| Drug-to-Antibody Ratio |
1.6
|
|||||
| Structure |
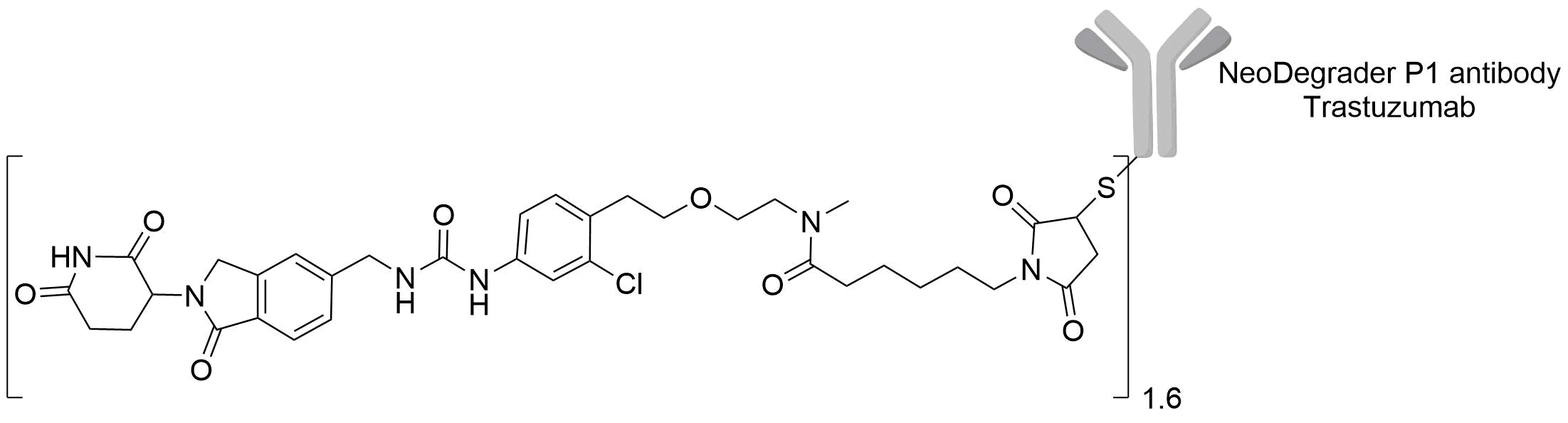
|
|||||
| Antibody Name |
Trastuzumab
|
Antibody Info | ||||
| Antigen Name |
Receptor tyrosine-protein kinase erbB-2 (HER2)
|
Antigen Info | ||||
| Payload Name |
NeoDegrader P1
|
Payload Info | ||||
| Therapeutic Target |
Protein cereblon (CRBN)
|
Target Info | ||||
| Linker Name |
Trastuzumab-Compound (ld) DAR 1.6 linker
|
|||||
General Information of The Activity Data Related to This ADC
Full List of Activity Data of This Antibody-drug Conjugate
Revealed Based on the Cell Line Data
| Experiment 1 Reporting the Activity Date of This ADC | [1] | ||||
| Efficacy Data | Half Maximal Inhibitory Concentration (IC50) | 74.00 pM | High HER2 expression (HER2+++) | ||
| Method Description |
Cells were plated at about 500 cells per well in a 96-well plate in 100 uL of media. In vitro activity and targeted delivery of ADCs, the isotype-matched negative controls ADCs, and naked antibodies control were assessed in cells.
|
||||
| In Vitro Model | Invasive breast carcinoma | BT-474 cells | CVCL_0179 | ||